|
They bombarded a uranium nucleus $^U$ following which the whole process repeats as described above, producing neutrons that are again bombarded with uranium nuclei, producing a perpetual chain reaction. This was essentially how nuclear fission was first discovered by Hahn and Strassman in 1939. Let us look at how this would work with an example of a Uranium atom bombarded by a neutron. Most reactions involve collision of a subatomic particle with an atomic nucleus that results in the formation of resultant fragments and particles. In controlled environments such as reactors, all nuclear fission occurs as a nuclear reaction that is bombardment-driven and involves collision of two subatomic particles. In any case, fission processes are exothermic reactions releasing energy in the form of electromagnetic radiation as well as kinetic energy imparted to the produced fragments. 
This process can occur naturally by spontaneous splitting of an atom via radiative decay or can be simulated in a lab by achieving appropriate conditions such as bombarding the heavy atom with subatomic particles like neutrons. The phenomenon of splitting up of a heavy atomic nucleus into lighter fragments and nuclei along with the emission of gamma rays or photons releasing large amounts of energy is termed as nuclear fission. As for the nuclear reaction, ensure that it is balanced at all stages. Remember that fission processes usually involve an exudence of a large amount of energy in various forms. In such a case, recall how this would be carried out in case of a uranium atom that is bombarded by a neutron, and the subsequent processes that follow through. As a result, an intense neutron flux and a fuel rich in fissile elements are both needed to compensate for this lower probability.Hint: We know that nuclear fission is a type of a nuclear reaction that involves splitting of atoms. The apparent cross-section of these targets is much more smaller for fast neutrons than it is for slower neutrons. 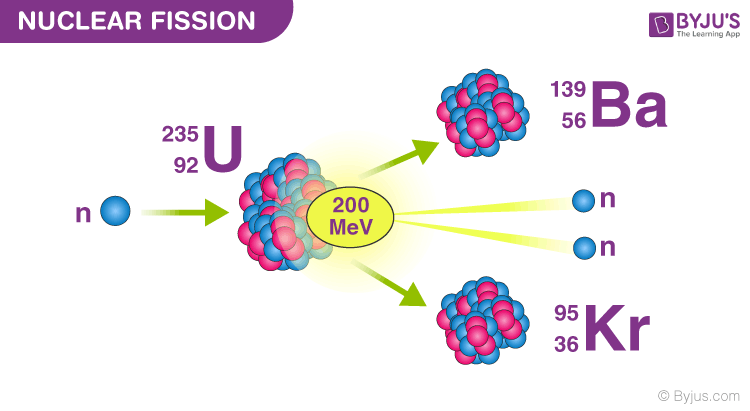
Travelling in matter, neutrons see nuclei as targets. One drawback of fast neutrons in reactors is that the probabilities of their capture by nuclei are comparatively small. This process is known as breeding, leading to an almost inexhaustible supply of nuclear fuel. The use of fast neutrons in so-called ‘fast reactors’ allows for the production of more fissile nuclei than are destroyed, as the absorption of at least one neutron per fission by an uranium 238 nucleus transforms this uranium 238 into a fissile plutonium 239 nucleus. Fast neutrons are needed to eliminate these waste products. They can therefore split not only nuclei reputed fissile by slow neutrons, but also minor actinides, the heavy nuclei which build up inside nuclear fuel as radioactive waste. The fact that they possess a substantial amount of kinetic energy allows fast neutrons to fission more easily nuclei once they get captured. They usually have energies between 0.1 and 2 or 3 MeV. Reactors operating with natural uranium fuel, which contains only 0,7% of fissile uranium 235, require efficient moderators which absorb very few of the neutrons : such moderators are heavy water and pure graphite.īefore they are slowed down by a large number of nuclear collisions, neutrons produced by fission reactions are known as 'fast'. 
As a result, the neutrons emitted by nuclear fission have to be slowed down by collisions within a medium called a moderator. Without them, the most common pressurised (PWR) and boiling water (BWR) reactors would not operate. It is these slow neutrons that allow for nuclear reactors to run with fuel based on natural uranium or uranium lightly-enriched in fissile isotope 235. Neutrons with energies in this range are collectively referred to as 'slow', and neutrons whose energies match those of the surrounding atoms are known as 'thermal'. Among these, some can poison the performance of the reactor, others are used for the control of the chain reaction (control rods).Īfter a series of collisions with different nuclei, the energy of neutrons produced by fission reactions drops to the order of a few electronvolts or a few fractions of an electronvolt. Other neutrons are lost when they are captured by 'sterile' nuclei or when they escape from the reactor as they slow down. Fast neutrons, on the other hand, play a small role in fission but can transform nuclei of uranium 238 into fissile plutonium 239. Slow neutrons are responsible for most of nuclear fission and therefore help sustain the chain reactions. This repartition of the neutrons in a pressurised water reactor shows the different roles played by slow and fast neutrons. Distribution of slow and fast neutrons in a PWR:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
